+91-11-45201100 drjindal@rdgc.in

Advance Hematology Analyzer
- Up to 110 samples per hour
- Autoloader can load 50 sample tubes
- Including NRBC with every CBC
- SF Cube three-dimension counting method for WBC 5-part, RET and NRBC, up to 37 reportable parameters and 29 research parameters
- 3 WBC channels contrastive analysis to get more accurate flag and WBC result for abnormal cells
- Optional Optical-PLT counting when large PLT or small RBC exists
- More reliable WBC result from avoiding the interference of NRBC
- Wide linearity range can count high value samples without dilution
- Both touch screen and PC operation to enhance convenience
- Recommended and customizable decision rules to re-exam abnormal samples
- Body fluid analysis, 7 reportable parameters and 11 research parameters
- Minimal aspiration sample volume , 80uL in whole blood mode,20ul in pre-dilute mode
- New working procedure brings efficiency by the software labxpert
- Single aspiration pathway
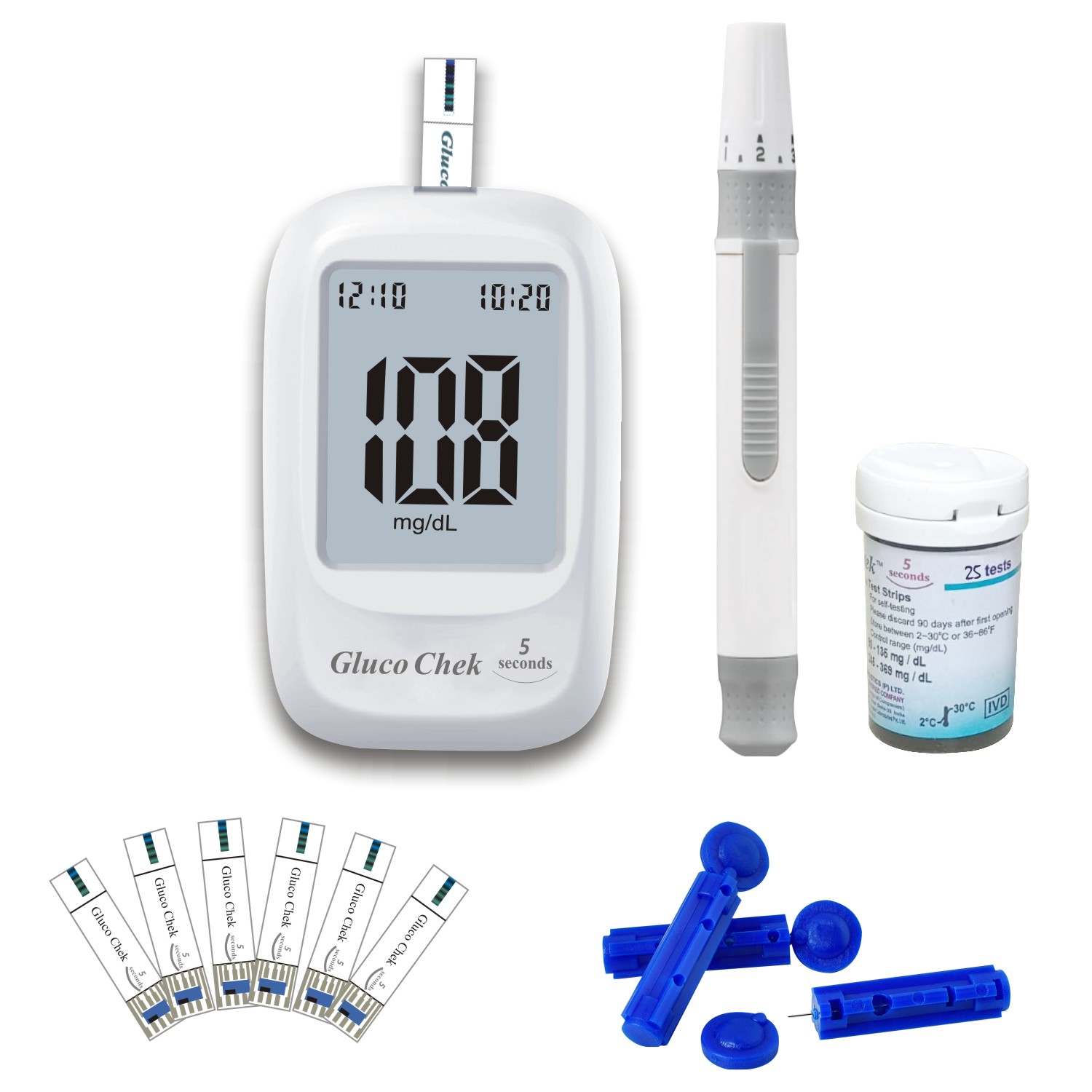
Aspen Gluco Chek 5 Seconds Glucometer
Aspen Gluco Chek 5 Sec Blood Glucose Glucometer Kit offers fast, accurate, and easy self-monitoring of blood glucose levels, helping you manage diabetes effectively. With a simple three-step operation and a hassle-free strip system, this glucometer delivers results in just 5 seconds using a small 0.5µL blood sample.
Product Features:
- Quick 5-second results for fast blood glucose monitoring
- Stores up to 1000 readings with date and time for easy tracking
- Requires only 0.5µL of blood sample for testing
- Simple three-step operation for user-friendly experience
- Hassle-free strip system for convenience
